Abortion, Without The Abortion Pill
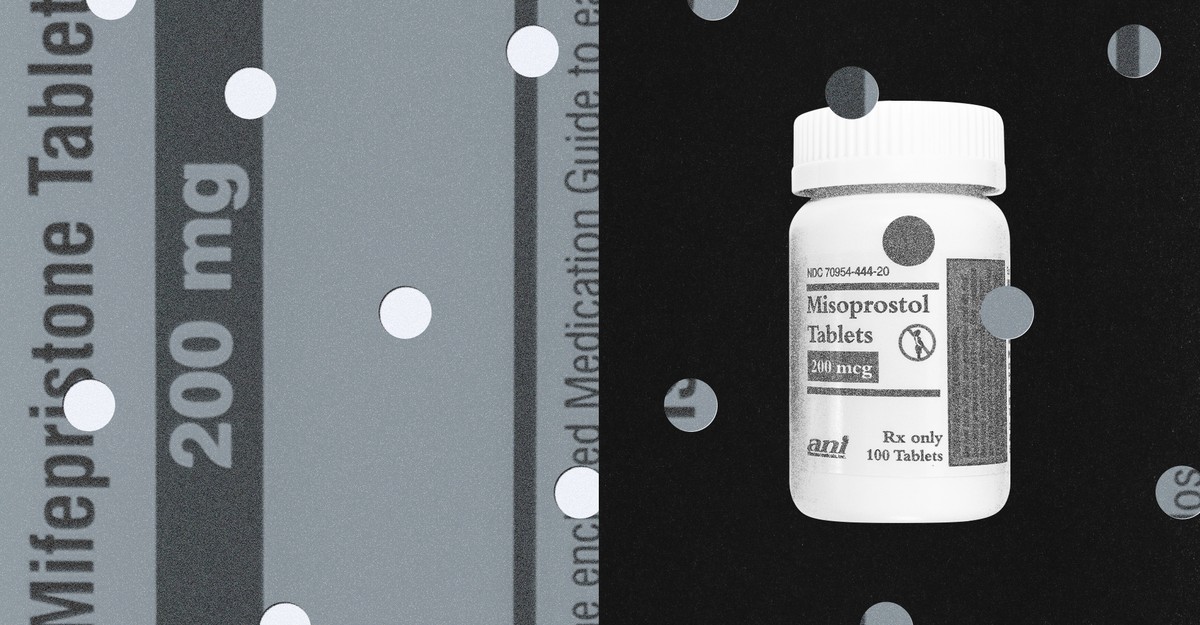
Over the weekend, reproductive-health-care providers across the country confronted a puzzle they had never before needed to solve at scale: how to offer medication abortion without mifepristone. The drug, also known as the abortion pill, is the first in a two-pill regimen that the FDA approved for pregnancy termination in 2000. Last Friday, the Fifth Circuit Court of Appeals blocked providers nationwide from prescribing it online or mailing it to patients, delivering the most sweeping shock to U.S. abortion policy since the overturning of Roe v. Wade.
Some abortion providers suspended telehealth services immediately. Others moved toward using only misoprostol, the second pill in the usual protocol, which can end a pregnancy on its own. Misoprostol-only abortion has long existed at the edges of American abortion care; now, for providers, it could serve as a strategic hedge against an unstable legal future.
Already, the Supreme Court has issued a one-week stay on the Fifth Circuit’s order, allowing mifepristone to once again be administered via telehealth. But mifepristone has been in the crosshairs of anti-abortion activists for as long as it’s been available in the United States. Even now, federal lawmakers are advancing legislation to ban mifepristone for medication abortion on the grounds that it is dangerous and likely to be abused. To assuage concerns about the potential for serious side effects, such as heavy bleeding and abdominal pain, the FDA long required doctors to prescribe the drug in person and supervise patients taking it. During the coronavirus pandemic, after reviewing data showing that patients could safely take the pills without an in-person clinical visit, the agency began allowing mifepristone to be prescribed via telehealth and delivered by mail.
Those changes were the focus of the case before the Fifth Circuit, a lawsuit in which the Louisiana government has argued that mail-order access to mifepristone has circumvented the state’s near-total abortion ban and that the FDA’s decision to remove the in-person dispensing requirements was based on flawed data. In court, the FDA has defended its current policy, but in September, the agency announced that it would revisit the drug’s prescribing rules. The Fifth Circuit’s Friday ruling barred telehealth prescription and mail delivery of mifepristone while that review proceeds.
[Read: A convenient piece of junk science]
If the ruling is upheld in the Supreme Court, it will affect a significant number of Americans. Medication abortion has been growing in use since its FDA approval and, as of 2023, accounts for nearly two-thirds of pregnancy terminations in the United States, according to the Guttmacher Institute, a nonprofit focused on sexual and reproductive health. About a quarter of all abortions are provided via telehealth. In states with abortion bans or heavy restrictions, receiving mifepristone and misoprostol by mail is one of the only paths to ending a pregnancy.
Misoprostol-only abortion is common around the world, especially in countries where abortion laws are restrictive or mifepristone is not widely available. “Of the two drugs, misoprostol was always the workhorse,” Heidi Moseson, a senior research scientist at Ibis Reproductive Health, an international research and advocacy group, told me. In the standard two-drug regimen, mifepristone is taken first to block progesterone, the hormone that helps sustain a pregnancy, and misoprostol follows 24 to 48 hours later, causing the uterus to contract in a process that mimics miscarriage. Taken alone, misoprostol yields the same result.
But in the United States, misoprostol-only abortion has generally been treated as a fallback option. This approach was built on studies published between 1994 and 2019 estimating the typical mifepristone-and-misoprostol regimen to be roughly 95 percent effective, compared with about 78 percent for misoprostol alone. The misoprostol-only approach was also thought to carry more side effects and a higher risk of incomplete abortion, which sometimes requires additional doses of abortion medication or a procedural abortion to resolve.
Recent evidence is more bullish on the efficacy and safety of misoprostol-only abortion. The studies included in the early reviews varied in dosage, timing, and route of administration of both mifepristone and misoprostol, making the two protocols difficult to compare directly. However, in 2023, the National Abortion Federation and the Society of Family Planning shared a standardized misoprostol-only protocol for medication abortion. A 2024 review of studies that followed this specific protocol found effectiveness rates ranging from 82 to 100 percent. Moseson, who led that review, is now helping lead the first randomized controlled trial to compare the two medication-abortion protocols. Recent research has also yielded mixed conclusions about whether misoprostol alone is associated with elevated rates of nausea, vomiting, and diarrhea, as was once believed. And serious complications are rare: Fewer than 0.2 percent of patients undergoing misoprostol-only first-trimester abortion require hospitalization or a blood transfusion. Studies in the United States and in countries where the misoprostol-only protocol is more common have found that patients can safely self-manage the regimen, even when they obtain misoprostol through online services rather than formal clinics.
[Read: The other abortion pill]
Stronger data on misoprostol-only abortion, Moseson told me, could expand the options available to patients. Misoprostol is typically cheaper and easier to access than mifepristone, including in states with abortion restrictions. Unlike mifepristone, which in the U.S. is primarily used for medication abortion, misoprostol can treat a variety of conditions, including stomach ulcers and postpartum hemorrhage. It has never been subject to the FDA’s special prescribing requirements and sits on retail-pharmacy shelves. (As of 2023, mifepristone can be dispensed by certified retail pharmacies, but in-store availability remains limited in practice.)
But for providers, rapidly moving more patients to a misoprostol-only protocol would not be simple. Many clinicians have less experience counseling patients through the regimen, including advising them on how to identify possible complications and confirm that an abortion is complete. New restrictions on mifepristone could also bring a fresh wave of scrutiny to abortion and reproductive health care more broadly, Lauren Ralph, an epidemiologist at UC San Francisco, told me. Ralph, who has studied the safety of telehealth and self-managed medication abortion, said she is not worried about the minimal medical risks of misoprostol-only abortion. The greater danger, she suggested, would be a legal one: In states with strict abortion laws, patients might delay seeking follow-up care or struggle to find a provider willing to risk criminal exposure by treating them.
Although untouched by the Fifth Circuit’s ruling, misoprostol is not necessarily immune to legal attacks. Louisiana’s lawsuit and parallel suits filed by Florida and Missouri argue that the Comstock Act—a dormant, 19th-century anti-obscenity statute that anti-abortion legal strategists have spent years working to resurrect—bars the mailing of any drug used to induce abortion. This reading would apply to misoprostol just as readily as to mifepristone. (The Fifth Circuit did not address that argument; whether the Supreme Court will do so remains an open question.) In 2024, Louisiana became the first state to classify both mifepristone and misoprostol as controlled dangerous substances, a designation that requires prescribing clinicians to acquire special licensure from the state and mandates that facilities store the drugs under lock and key. All prescriptions must be tracked through a state database, and anyone possessing the pills without a valid prescription faces fines or jail time. Lawmakers in Texas, Missouri, and Kentucky have proposed copycat bills.
[From the January 2024 issue: A plan to outlaw abortion everywhere]
The places least equipped to absorb new storage and prescribing requirements are the same places where access to health care is already most precarious. Michelle Erenberg, the executive director of the reproductive-rights organization Lift Louisiana, told me that some rural and small community hospitals—which typically have limited capacity to store controlled substances—have effectively lost access to misoprostol for any kind of care. Pharmacists across the state have stopped stocking the drug, or grown wary of filling prescriptions, she said, leaving patients in limbo as they seek misoprostol for outpatient miscarriage care or to prepare for procedures such as IUD insertion. In December 2024, two months after Louisiana’s law took effect, a doctor performing an emergency C-section preordered misoprostol—previously kept within arm’s reach on delivery-room trays—in anticipation of a possible hemorrhage. Yet when the patient began losing blood on the table, the medication was still in a locked pharmacy cabinet across the hospital.
Abortion providers can adapt to mifepristone’s absence, as many already have. Misoprostol, by contrast, is harder to replace, as a mainstay of obstetric care and the most viable alternative to the standard regimen for pregnancy termination. If it becomes widely restricted, Americans seeking abortion care will have no ready substitute.
Popular Products
-
 Ashwagandha & Black Cumin Seed Oil
Ashwagandha & Black Cumin Seed Oil$43.56$21.78 -
 Shilajit Capsules for Strength & Vita...
Shilajit Capsules for Strength & Vita...$43.99$29.78 -
 Pre-Workout Supplement for Strength, ...
Pre-Workout Supplement for Strength, ...$43.99$29.78 -
 Centrum Multivitamin for Energy, Immu...
Centrum Multivitamin for Energy, Immu...$43.99$25.78 -
 Bee Venom Capsules for Joint Support ...
Bee Venom Capsules for Joint Support ...$43.56$21.78